Gene therapy’s quiet pivot to precision
A six-week data snapshot shows clinical activity building where press attention isn’t - three companies, thirteen trials, and fifteen orphan designations.
FDA sets PDUFA date for Telix’s TLX101-Px glioma imaging
September 2026 PDUFA date given to Telix’s glioma imaging agent.
NICE positions semaglutide in cardiovascular prevention
Guidance expands semaglutide use beyond weight management into cardiovascular prevention.
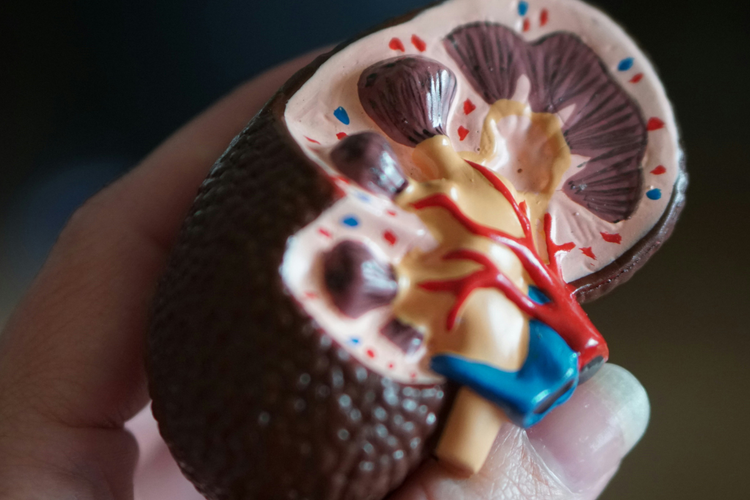
Filspari wins first FDA nod for FSGS Proteinuria
Sparsentan cut proteinuria 46% versus 30% for irbesartan at 108 weeks in Phase III DUPLEX, the largest FSGS trial to date, but missed its primary kidney function endpoint.
Nxera hits second Lilly milestone in metabolic deal
Nxera's NxWave platform has now delivered two development milestones to Lilly within ten months, signaling recurring delivery
AtaiBeckley's BPL-003 shows rapid, durable TRD response on SSRIs
Single intranasal dose of BPL-003 achieved 66.7% antidepressant response by Day 2 in treatment-resistant depression patients on stable SSRIs, with 83% durability at Day 85.
Lilly, Novo, and the new terms of the oral obesity market
Orforglipron outperformed Rybelsus on every efficacy measure. The discontinuation rate is the number that matters now.